- Blood pressure: 40/0 mmHg
- Heart rate: 112 b/min
- Respiratory rate: 25/min
- SpO₂: 89%
- NT-pro-BNP=465 pg/mL
- HsTrop I=25 ng/mL
- HB= 1.37 g/dl
- sCreat=1.61 mg/dl
- CRP 2.24 mg/mL
- ALT 42.7 ME
- AST 243.4 ME
- Two stents into the distal left main and the proximal LAD.
- From the distal RCA into the proximal PDA.
- NT-proBNP: 35,000 pg/mL
- Procalcitonin (PCT): 3.63 ng/mL
- Hemoglobin (Hb): 8.9 g/dL*
- Serum creatinine: 2.0 mg/dL
- C-reactive protein (CRP): 10.0 mg/mL
- ALT: 19 U/L
- AST: 31.5 U/L
- BUN: 24.3 mg/dL
- AR minimal
- MR severe, central jet (VC 0.9)
- TR severe
- LVEF = 18%, EDV = 126ml, ESV = 103ml
- Pericardial effusion ↑ from mm to 25 mm
- TAPSE 9 mm
- S’ medial 4 m/s
- S’ lateral 4.2 m/s
- IVC 23 mm
- Akinesis of the anterior, anterior septal, anterolateral walls mainly at the level of the middle and apical segments. Akinesis of the apical segments
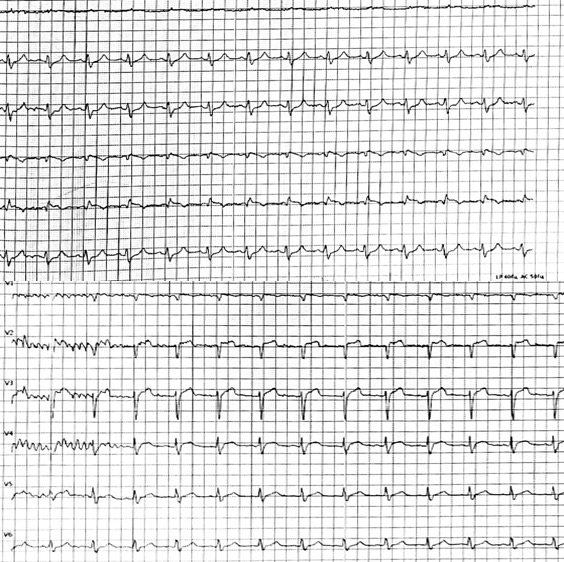
A 63-year-old male (90 kg; BMI 28 kg/m²) with a history of hypertension presented with acute chest pain that began approximately two hours prior to admission.
Pre-hospital assessment (ambulance):
He was transferred for urgent coronary angiography upon arrival to the emergency department.
Urgent blood tests:
Angio
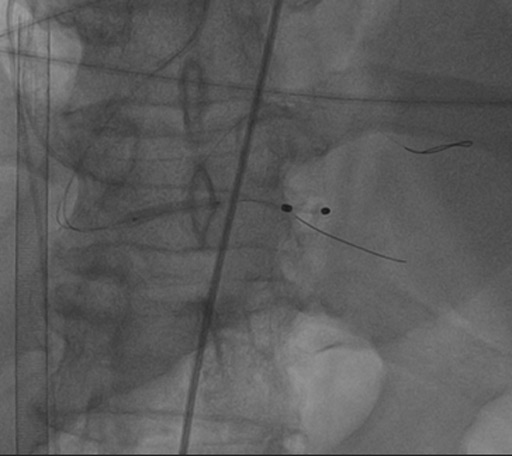
Coronary angiography revealed severe multivessel coronary artery disease, including LM occlusion, severe distal RCA occlusion, and 40% proximal PDA stenosis. During engagement of the guiding catheter to the LAD, the patient developed ventricular fibrillation. Successful resuscitation was achieved within 5 minutes. Veno-arterial ECMO was initiated at a flow of 2 L/min, followed by intra-aortic balloon pump (IABP) implantation.
Three drug-eluting stents were implanted:
Wiring the LAD
First stent was implanted
We see a contrast stagnation after the LAD stenting
The next stage was of RCA distal part stenting.
Following weeks
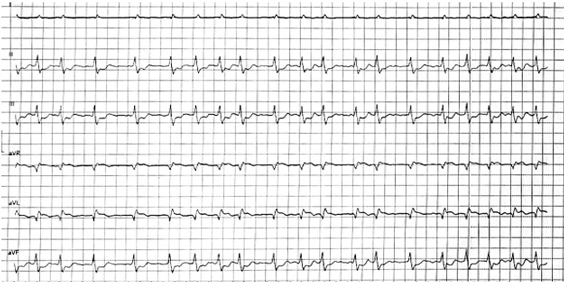
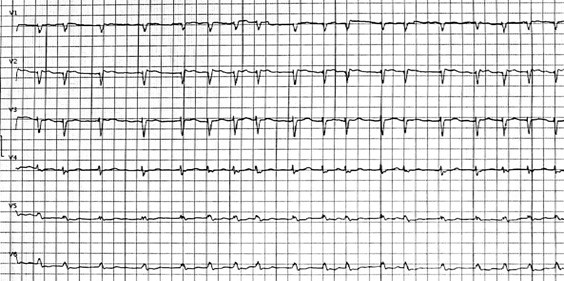
VA-ECMO and IABP were weaned and explanted using a stepwise approach.
The patient subsequently developed atrial fibrillation. Ultrafiltration therapy was initiated.
Laboratory findings showed:
- Severe symptomatic MR (NYHA IV) and high prohibitive surgical risk (Class IIA recommendation, level of evidence B-NR )
- Patient underwent Mitraclip placement medially to A3 P3; Following the postoperative period, an echocardiogram showed a reduction MR severity from severe to mild with mean MV gradient of 3.6 mmHg.
- Mechanical Support (ECMO/IABP):Was discontinued prior to MTEER (ECMO on 11/02/2025, IABP on 14/02/2025). No new mechanical support was required after MTEER. However, even after Mitraclip procedure, the inotropic and vasopressor support was required using Norepinephrine 0.12 mcg/kg/min, Dobutamine 7 mcg/kg/min, Vasopressin 0.03 U/min during hospitalization.
Our patient met the criteria for transcatheter mitral repair:
- Mechanical circulatory support decision-making
VA-ECMO and IABP play complementary roles in stabilizing profound cardiogenic shock, improving perfusion, and enabling staged diagnostics. - Identification of organ dysfunction and timing of interventions
Rising creatinine, BUN, CRP, and NT-proBNP should prompt early renal support—including ultrafiltration—to optimize volume status. - Atrial fibrillation in advanced HF
New-onset AF after ECMO weaning is common and can significantly worsen hemodynamics; early rate/rhythm control is pivotal. - Revascularization and surgical choices
In high-risk multivessel CAD, complete revascularization remains essential for survival. - Management of ischemic mitral regurgitation after STEMI
Severe ischemic MR after large MI significantly worsens prognosis and requires timely multidisciplinary evaluation.
Functional MR may improve with revascularization and unloading, but persistent severe MR often necessitates intervention.
Stepwise weaning is essential to avoid hemodynamic collapse and uncover residual LV dysfunction or valvular pathology.Edge-to-edge mitral repair (TEER) is an emerging option for patients at high surgical risk, whereas surgery remains standard when papillary muscle rupture is suspected.
This case illustrates the extreme end of the cardiogenic shock continuum, where rapid deterioration, multiorgan dysfunction, and evolving mechanical complications intersect to challenge clinical decision-making. The patient presented in profound shock with LM occlusion, evolving multisystem injury, and subsequently developed severe ischemic mitral regurgitation—an often under-recognized driver of hemodynamic collapse after large anterior myocardial infarction.
The management demonstrates a well-coordinated and multidisciplinary staged strategy: early ventricular fibrillation arrest was rapidly reversed, and veno-arterial ECMO with IABP provided circulatory support that allowed complete revascularisation and stabilisation. This combination—ECMO for systemic perfusion and IABP for LV unloading—remains a pragmatic approach when shock physiology is severe and time-critical. Alternatively, the combination of ECMO and Impella (ECPELLA) may offer enhanced ventricular unloading, representing an emerging synergistic model in similar scenarios.
Despite mechanical support and successful PCI, the persistence of severe MR underscores the dynamic interplay between infarct size, papillary muscle dysfunction, and tethering of the mitral apparatus. In such cases, MR is not merely a bystander but a major amplifier of LV failure and could ultimately lead to cardiogenic shock and multiorgan failure. The decision to pursue transcatheter edge-to-edge repair was appropriate given the prohibitive surgical risk and the patient’s ongoing symptomatic burden. TEER offers a valuable option for post-MI MR when surgical intervention is unsafe, and this case reinforces the importance of early multidisciplinary evaluation.
Equally instructive is the evolution of organ dysfunction—renal impairment, inflammatory activation, atrial fibrillation—each of which complicates recovery and highlights the need for close monitoring during ECMO support and weaning. Ultimately, this case exemplifies contemporary cardiogenic shock management: early mechanical support, complete revascularization, identification of residual mechanical pathology, and thoughtful application of transcatheter therapies to restore stability and improve prognosis.
None
Disclaimer
This case report and/or content does not reflect the opinion of iHF or iheartfunction.com, nor does it engage their responsibility.






(No subject)